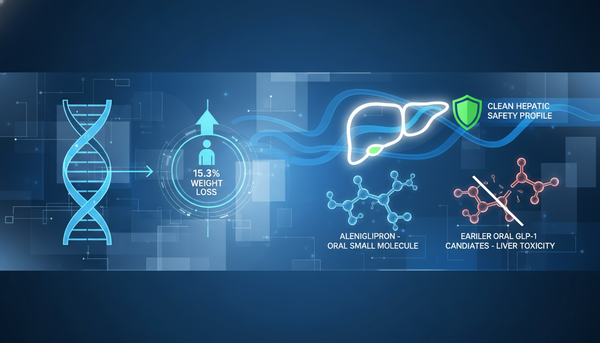
Oncology
Clinical Breakthrough at ASH 2025: CLN-049 Demonstrates 50% Response Rate in TP53-Mutated AML via Novel FLT3xCD3 Engagement
Phase 1 data confirms extracellular FLT3 binding efficacy regardless of mutational status, offering a potential breakthrough for refractory subsets. * These analyses reflect my personal opinions and may include input from multiple sources. They are for informational purposes only and do not constitute professional advice. * Clinical Perspective Data presented today at